This artist's impression shows a qubit (purple arrow near center) in a silicon carbide crystal interacting with nuclear spins that are derived from naturally occurring silicon-29 (green arrows) and carbon-13 (red arrows) isotopes. Image courtesy of Hosung Seo, Institute for Molecular Engineering.
by Benjamin Recchie
Quantum mechanics has been upending our understanding of the universe for a century. Particles blink into existence and just as quickly vanish. Light behaves as a wave and as a particle at the same time. Add to these examples results from a new paper in Nature Communications by postdoctoral scholar Hosung Seo (who works with Giulia Galli, Liew Family Professor of Molecular Engineering, the University of Chicago) and Abram Falk (formerly of the research group of David Awschalom, Liew Family Professor in Molecular Engineering, the University of Chicago, but now at IBM’s T.J. Watson Research Center in Yorktown Heights, New York), in which a material being investigated for applications in quantum computing exceeded all expectations. The other coauthors on the paper are.graduate student Kevin Miao and former graduate student Paul Klimov (now at Google, Inc. in Santa Barbara).
Quantum computers, like electronic computers, manipulate information in bits. But while an electronic computer stores bits as a 1 or a 0, a quantum bit (or qubit) can store information in a superposition of many states at once. This unique property makes quantum computing highly desirable for certain classes of difficult-to-solve problems, such as simulating quantum mechanical systems in chemistry, or breaking codes.
One of the great challenges of the attempt to build a practical quantum computer has been decoherence—the process by which the qubit decays and its information is lost irretrievably. This typically takes far less than a millisecond to happen in the solid-state, so many researchers are working to discover ways to keep the qubit coherent long enough to be usable. Some researchers have tried to overcome this by cooling their qubits to almost absolute zero, an expensive and complex undertaking; others, such as those in the Institute for Molecular Engineering, are trying to identify semiconductors with properties that can hold a qubit under less extreme conditions.
As it turns out, there is a common, well-understood material that’s known to hold a qubit—diamond. A diamond is a crystal made entirely of carbon; swap one carbon in the crystal lattice with a nitrogen atom, and you set up an imperfection in the crystal, called a nitrogen vacancy, that can hold a qubit. But pure diamond isn't an ideal material for quantum computing, explains Falk. Not only is it expensive and hard to work with, but it's hard to transmit quantum information very far within it with light, which constrains its uses. Instead, the researchers turned to silicon carbide (SiC), since there’s already a well-developed industry built around industrial SiC, the raw materials are cheap, and it's easier to transmit light through it. The Awschalom group figured that removing one silicon and one carbon atom from the crystalline matrix would create a divacancy, a two-ion-sized hole that could hold a qubit. In laboratory tests, SiC performed well—in fact, too well. The researchers managed to keep the qubit coherent for 1.3 ms, the longest coherence time yet recorded in a natural crystal, and much longer than they expected. And like all scientists, they wanted to know more about how their good fortune came about.
The researchers turned to postdoctoral scholar Hosung Seo for help. Seo has been using high-performance computing to design custom materials for quantum computing applications as part of his research with the Galli group. The Awschalom group asked him to work backwards and help them find out why SiC was performing so well. Seo developed a custom code to study the problem and ran it on Midway, the Research Computing Center’s high-performance computing cluster.
What the researchers discovered was that it was the binary nature of the crystal itself that delayed the decoherence of the qubit. In other semiconductors, a major source for the decoherence of the qubit is created by the spin (a quantum characteristic of all particles) of the other atoms’ nuclei. The spin of the protons and neutrons in each carbon atom created tiny magnetic fields, which created a kind of “noise.” But while most carbon atoms—called 12C—have a total of 12 protons and neutrons, a small fraction—called 13C—have an extra neutron. The mismatch in the spins between the two carbon isotopes creates noise that destabilizes the qubit in less than a millisecond in diamond.
Common sense would suggest that SiC, which contains two different elements, would have even more mismatched spins and thus more noise. But common sense doesn’t always apply in the quantum world. In fact, Seo found that that the differences in the magnetic constants of the spin from the two elements suppressed the noise that destroyed the information within the qubit, in effect creating a sheltered place for the qubit to stay coherent. Furthermore, the greater distance of carbon atoms from each other and silicon atoms from each other in the lattice meant that each element’s spin-created noise affected the qubit less than if the atoms were all the same element.
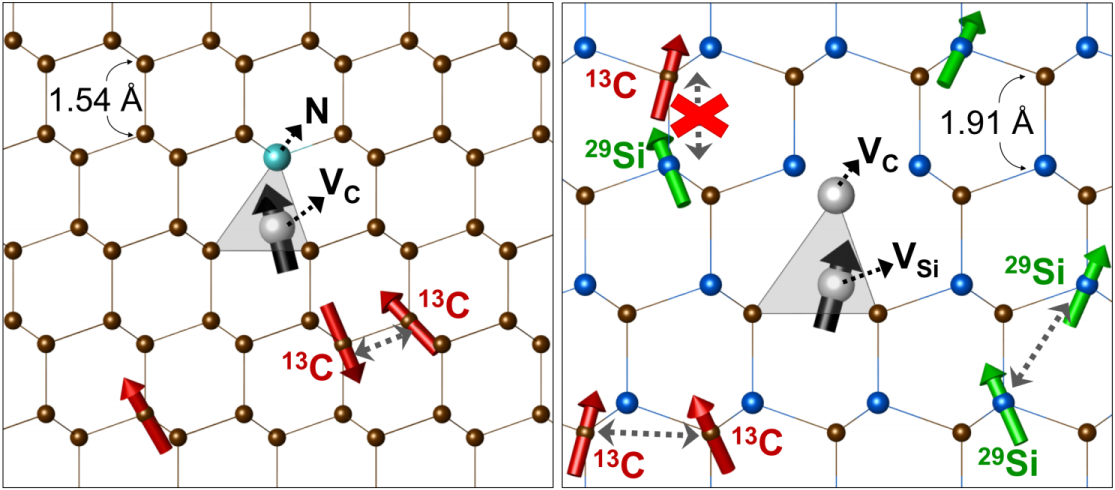
This diagram shows (left) a nitrogen vacancy in a diamond lattice, and (right) a divacancy defect complex in silicon carbide. Spins from carbon-13 and silicon-29 are marked. Note the greater distance between atoms in the lattice in the silicon carbide--part of the reason the silicon carbide allows for longer-lived qubits. Courtesy of Hosung Seo.
The discovery of the suppressed noise opens up a new frontier for exploring materials suitable for quantum computing, Seo and Falk say; researchers can focus on complex crystals made from two, three, or even more elements. Seo and Falk believe that host crystals with carbides, nitrides, and oxides are particularly promising.
“The beauty of this theoretical work is that it’s basically based on a really general model,” says Seo. “The physics of this can be applied to any other qubits in any other solid material.” In other words, this finding could have a big impact for the field in the not-too-distant future.